
The mycelium
While we may admire a mushroom growing out from the soil or a bracket fungus growing out from a log, it is the out-of-sight (and often forgotten) mycelium that is the essential part of the organism. The mycelium is there throughout the year, in the soil or in the log, and is not a static object. It grows and may die. It reacts to varying environmental conditions and other organisms, producing different growth forms or structures, depending on circumstances.
This section will discuss some of the features of the mycelium.
Hyphal growth
Consider a spore from a simple mushroom, out in a field. The spore germinates
and produces a short, initial hypha (called a germ tube). The germ tube
grows and branches, each of those early branches grows and branches in turn
and the process continues. The following diagram shows this process. 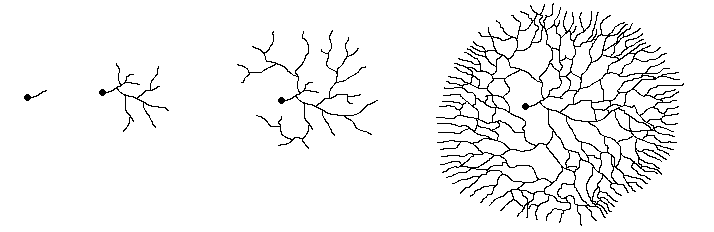
The leftmost figure shows a spore (the black dot) with the short germ tube growing
out from it. The next figure shows the scene a little later, with several branchings
having occurred by now. The other two figures show later stages in the expansion
of the mycelium. Notice how, by repeated branching, the mycelium eventually
assumes a circular form as shown in the rightmost figure. That figure also shows
that while the hyphae show a very marked outward growth, there are also cross-connections
between the outward growing branches. The cross-connections between the radiating
hyphae make it easy to move nutrients quickly around the growing mycelium, taking
them to wherever they are most needed.
This website deals with the macrofungi and so microfungi such as the various moulds aren’t included. However, it’s worth noting that the common household moulds on bread and fruit provide excellent examples of the radial growth that has just been explained. These mould colonies are also composed of hyphae and are far more easily seen than the out-of-sight mycelium of a mushroom. Radial growth is a feature common to all fungi (macro and micro) that grow as hyphae. The yeasts are examples of non-hyphal microfungi, being single-celled and budding off daughter cells.
In many hyphal species, the individual hyphae in a mycelium are no more than
a hundredth of a millimetre in diameter. A hypha grows through the addition
of new material to the hyphal wall at or near the tip of the hypha and this
reference button goes into technical detail about the mechanics of tip growth.
A hypha grows by increasing in length. There is no increase in diameter (as
in a plant root for example) but, as shown in the diagram above, hyphae branch.
![]() Branching
normally occurs some distance behind the tips, so that the margin of a growing
mycelium is typically composed of unbranched leaders, growing into fresh territory.
Branching
normally occurs some distance behind the tips, so that the margin of a growing
mycelium is typically composed of unbranched leaders, growing into fresh territory.
The hyphae are not just uniformly featureless threads, the same in all species.
For example, the hyphae may be smooth or encrusted, colourless or coloured and
the hyphal walls may be two-layered to multi-layered. The hyphae of the macrofungi
(and numerous microfungi) have cross-walls (or septa), which give structural
support - and there are variations in septal structure. There are various chemical
components to the hyphal walls, with chitin a significant component in the macrofungi.
Insect exo-skeletons are also composed of chitin. While the prime purpose of
a hyphal wall is to keep the inside in and the outside out, that need not be
its only role. It can also be used as a place to store food reserves. For example,
when it comes to producing fruiting bodies, the species Schizophyllum commune
![]() breaks down parts of the hyphal walls in the mycelium and uses the released
nutrients in fruit body production.
breaks down parts of the hyphal walls in the mycelium and uses the released
nutrients in fruit body production.
In order to feed, the mycelium releases enzymes into the surrounding environment and these enzymes break down complex organic polymers into simpler compounds (generally various sorts of sugars) which are then absorbed through the hyphal walls. All this also happens near the growing hyphal apices, generally just behind the region where fresh material is being added to the hyphal wall. While this discussion uses a common mushroom as a simple example, it’s important to note that this method of feeding - enzyme release into the surrounding environment and absorption of the resulting products through the cell walls - is a fundamental characteristic of all fungi, whether macrofungi or microfungi.
Hyphal growth need not be random, since fungi respond to chemical signals. For example the hyphae of various parasitic or mycorrhizal species respond to chemicals that leak out from the tips of plant roots.
 The
combination of generally radial growth, with branching hyphae growing out from
behind the leading hyphae, means that a mycelium can explore and exploit a large
area. As the mycelium exhausts the food sources in one area and expands outward
in a circular fashion, there's no benefit in maintaining the inner mycelium.
So the fungus cannibalizes the inner mycelium, extracting whatever nutrients
it can from there and moves them to the outer, growing regions. Whatever cannot
be recycled is shut off from the growing region and allowed to decay, so that
the mycelium actually grows as an expanding ring, not as an expanding disk,
and the accompanying diagram shows this.
The
combination of generally radial growth, with branching hyphae growing out from
behind the leading hyphae, means that a mycelium can explore and exploit a large
area. As the mycelium exhausts the food sources in one area and expands outward
in a circular fashion, there's no benefit in maintaining the inner mycelium.
So the fungus cannibalizes the inner mycelium, extracting whatever nutrients
it can from there and moves them to the outer, growing regions. Whatever cannot
be recycled is shut off from the growing region and allowed to decay, so that
the mycelium actually grows as an expanding ring, not as an expanding disk,
and the accompanying diagram shows this.
If you've seen mushrooms growing in fairy rings in a field, this ring-like mycelial growth immediately suggests the cause. That is in fact the case, but one important point is that, generally speaking, the mycelium produced from a single spore will not produce fruiting bodies. It is necessary for two mycelia to mate and the resulting, expanding mycelium can then produce the fairy-ring mushrooms.
The earlier illustration of radiating mycelial growth showed a somewhat diffuse growth pattern, with individual hyphae growing out in all directions. If there is a fairly uniform distribution of nutrients in a benign environment, such radial growth is very efficient since it allows the fungus to make the most of those nutrients. However, nutrients are not always uniformly distributed and the surrounding environment is often hostile. These factors are major influences on the pattern of mycelial growth. If there's a significantly higher concentration of nutrients in one area, then the mycelium would grow preferentially into that area, with less growth (or perhaps even none) into other areas - especially areas with a hostile environment.
In an area rich in nutrients, the mycelium will branch often and grow slowly, so maximising the amount of nutrients it can extract. By contrast, in an area low in nutrients the hyphae grow more rapidly and with little branching. Moreover, in many cases the bulk of the mycelium doesn't grow as individual hyphae - but as bundles of hyphae. Some bundles are simply fairly loose and unstructured aggregations of hyphae, with no differentiation in the functions of the component hyphae. The opposite also occurs commonly, with a definite structure to the bundle and marked differentiation in the nature of the hyphae making up the bundle.
Rhizomorphs
These hyphal bundles are referred to by various terms (mycelial cords,
mycelial strands or rhizomorphs) and you will see some variation
in the use of these terms. Some people use the term rhizomorph for anything
but the loosest of bundles whereas others will use it only for highly structured
bundles. I'll use the word "rhizomorph" here as a general term for all such
bundles. ![]() The
reference given in the button contains a discussion of some of the terms that
have been used for such hyphal bundles.
The
reference given in the button contains a discussion of some of the terms that
have been used for such hyphal bundles.
The photo ![]() shows some white rhizomorphs growing through an area of wood chip mulch. You’ll
often come across such rhizomorphs when raking aside dead wood or old mulch,
while doing some gardening. There is considerable variation in both the structure
and growth of rhizomorphs. In some species the rhizomorphs grow in a very regimented
way, the tips extending as a whole, in a manner similar to the growth of root
tips in plants. In other cases one or a few founder hyphae grow out and
other hyphae then follow the same route as the founder hyphae and twine around
them. In this way a bundle of hyphae builds up. In many species the outer regions
of the mycelium grow in a diffuse, fanned-out fashion with rhizomorphs developing
a little behind those expanding, diffuse extremities.
shows some white rhizomorphs growing through an area of wood chip mulch. You’ll
often come across such rhizomorphs when raking aside dead wood or old mulch,
while doing some gardening. There is considerable variation in both the structure
and growth of rhizomorphs. In some species the rhizomorphs grow in a very regimented
way, the tips extending as a whole, in a manner similar to the growth of root
tips in plants. In other cases one or a few founder hyphae grow out and
other hyphae then follow the same route as the founder hyphae and twine around
them. In this way a bundle of hyphae builds up. In many species the outer regions
of the mycelium grow in a diffuse, fanned-out fashion with rhizomorphs developing
a little behind those expanding, diffuse extremities.
 The
following two diagrams will give you some idea of the features found in many
of the highly structured rhizomorphs, with well regimented tip growth. But remember
that there is variation in rhizomorph structure, so these are generalized diagrams,
rather than faithful depictions of the rhizomorphs of any particular species.
The
following two diagrams will give you some idea of the features found in many
of the highly structured rhizomorphs, with well regimented tip growth. But remember
that there is variation in rhizomorph structure, so these are generalized diagrams,
rather than faithful depictions of the rhizomorphs of any particular species.
The first diagram represents a cross-section, taken along a rhizomorph. The
growing apex is on the right, where you can see the individual hyphae, intertwined
but generally parallel and, in this area, the hyphae show no differentiation
in function. A little way further back there is a marked difference in structure
across the rhizomorph. Moving from the outside to the centre:
 The
narrow black outer zone represents an outer skin - solid and black in a number
of species, but not always
The
narrow black outer zone represents an outer skin - solid and black in a number
of species, but not always
The dark brown area represents a region packed with narrow hyphae
The speckled blue/light brown area contains many wider hyphae
The second diagram shows a cross-section taken along the red line, well behind the growing apex, in the first diagram. Once again you can see the thin, black protective skin and just in from that the band of narrow hyphae. The area that was a speckled blue/brown in the previous diagram is now shown in more detail, with a mix of narrow and broad hyphae (the hyphal walls shown in blue and the interior white). The broad hyphae are called vessel hyphae and the interiors of these are often empty, allowing the easy flow of nutrients. Within the broad central region the light brown area may contain more narrow hyphae or be composed of glues that bind the hyphae together.
As an example of the variation in rhizomorph structure, the Australian species
Armillaria luteobubalina ![]() produces rhizomorphs where there is a very large, mostly empty channel constituting
a large part of each rhizomorph. It had long been supposed that this channel
would allow the efficient transport of oxygen to the advancing apex, but proof
of this has come only fairly recently. Laboratory experiments at the University
of Sydney and the University of New South Wales have shown that rhizomorphs
of Armillaria luteobubalina conduct oxygen. When this species was grown
on agar in a petri dish, cylindrical air pores grew vertically above the fungal
culture, to about 7mm in height. Rhizomorphs developed below the air pores,
grew a short distance down into the agar and then turned to continue growing
horizontally. The channels within the air pores were continuous with the central
channels in the rhizomorphs and tests showed that oxygen moved through the system.
This reference-button takes you to an abstract of a talk about this work, given
at the 2002 International Mycological Congress in Oslo. A couple of the people
engaged in this work found air-pore-like structures in Norwegian Armillaria-infected
wood, during a field excursion associated with the Congress.
produces rhizomorphs where there is a very large, mostly empty channel constituting
a large part of each rhizomorph. It had long been supposed that this channel
would allow the efficient transport of oxygen to the advancing apex, but proof
of this has come only fairly recently. Laboratory experiments at the University
of Sydney and the University of New South Wales have shown that rhizomorphs
of Armillaria luteobubalina conduct oxygen. When this species was grown
on agar in a petri dish, cylindrical air pores grew vertically above the fungal
culture, to about 7mm in height. Rhizomorphs developed below the air pores,
grew a short distance down into the agar and then turned to continue growing
horizontally. The channels within the air pores were continuous with the central
channels in the rhizomorphs and tests showed that oxygen moved through the system.
This reference-button takes you to an abstract of a talk about this work, given
at the 2002 International Mycological Congress in Oslo. A couple of the people
engaged in this work found air-pore-like structures in Norwegian Armillaria-infected
wood, during a field excursion associated with the Congress. ![]() Those
structures, produced by a European species of Armillaria, looked very
much like the air pores produced in the laboratory experiments with the Australian
Armillaria luteobubalina.
Those
structures, produced by a European species of Armillaria, looked very
much like the air pores produced in the laboratory experiments with the Australian
Armillaria luteobubalina.
Armillaria is a world-wide genus of parasitic fungi, with different
species in different continents and the species of this genus produce well-structured
rhizomorphs. Vessel hyphae have been found in the rhizomorphs of some Armillaria
species, but not in Armillaria luteobubalina. The researchers at the
universities of Sydney and New South Wales point out that this does not mean
that Armillaria luteobubalina rhizomorphs don’t conduct nutrients
- just that they don’t have empty vessel hyphae to do the job. ![]() In
this species, the main role of the rhizomorphs is the transport of oxygen through
areas with low oxygen levels - such as moist soils.
In
this species, the main role of the rhizomorphs is the transport of oxygen through
areas with low oxygen levels - such as moist soils.
Apart from various parasitic fungi, such as Armillaria, many saprotrophic
and mycorrhizal species also produce rhizomorphs. The photo ![]() shows two immature fruiting bodies of the endemic Australian mycorrhizal species
Dermocybe globuliformis, revealed when the surface layer of leaf litter
was pulled aside. You can see some diffuse pale yellow mycelium as well as a
few, darker yellow rhizomorphs.
shows two immature fruiting bodies of the endemic Australian mycorrhizal species
Dermocybe globuliformis, revealed when the surface layer of leaf litter
was pulled aside. You can see some diffuse pale yellow mycelium as well as a
few, darker yellow rhizomorphs.
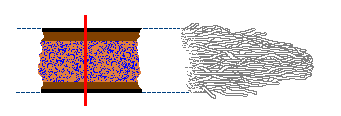
Rhizomorphs serve varied purposes. The example of oxygen transport in Armillaria has already been mentioned. Many rhizomorphs can also be looked at as exploratory or migratory structures. For example, suitable food sources for saprotrophic, wood-decay fungi can be widely separated from each other. Look at the fallen trunks or branches in a woodland. Typically there is some distance between them. To you the distances between the different bits of fallen timber may not seem great, perhaps a half-metre here or a few metres there, but to a fungus the intervening areas of bare, wood-free soil may as well be oceans. If such a fungus has exhausted all the nutrients in one piece of wood, it faces a challenge getting to another piece of wood. Rhizomorphs can offer considerable advantages as exploratory tools. The outer skin protects the inner hyphae in the traversal of an inhospitable region. Additionally, rhizomorphs grow much more quickly than isolated hyphae, thereby speeding up the exploration for fresh food sources.
Rhizomorphs also offer considerable advantages when a new piece of wood is found. As there are typically numerous hyphae making up a rhizomorph, when it strikes a new piece of wood the numerous hyphal tips can fan out and rapidly colonise the new wood as a diffuse, branched mycelium. Compare the two figures in the diagram below. In each case the brown rectangles represent pieces of wood. In figure 1 a rhizomorph has come from the left and found the wood, whereupon the constituent hyphae fan out into a radiating mycelium and fairly quickly have occupied a good proportion of the wood. Figure 2 shows what happens when just a single hypha strikes a similar piece of wood. Once in the wood it will also branch and spread out, but for any time period it will have occupied less of the wood than the rhizomorphic fungus.
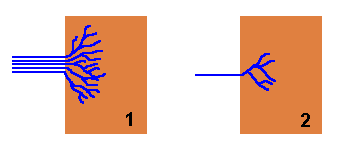
A well-structured rhizomorph that is able to transport large amounts of water, nutrients or oxygen to the advancing apex will easily fuel the rapid expansion of the fungus into new territory. The contrast between a rhizomorph and an individual hypha is similar to the difference between a multi-lane highway and a narrow, winding road.
If the new wood is unoccupied by any other fungi or micro-organisms, figure
1 shows that the newly arrived fungus can quickly colonize this pristine
territory before it's found by other organisms. On the other hand, the new piece
of wood may already be occupied by other fungi or various micro-organisms, in
which case the newly arrived fungus must contend with those existing occupants.
If the new fungus has arrived via a rhizomorph then it may be able to overcome
the defences of the existing occupants because of the mass of hyphae that are
brought to the wood simultaneously. To use a military analogy, ![]() figure
1 is equivalent to landing an army division in enemy territory whereas
figure 2 is equivalent to landing only a small squad of soldiers in the
same hostile territory.
figure
1 is equivalent to landing an army division in enemy territory whereas
figure 2 is equivalent to landing only a small squad of soldiers in the
same hostile territory.
Armillaria luteobubalina is one example of rhizomorphs enabling a fungus
to traverse inhospitable areas. A further example of this is the Dry Rot fungus
(Serpula lacrymans ![]() ), a species that can cause significant damage to timber in damp, poorly ventilated
buildings. The rhizomorphs produced by this fungus can extend for several metres
across brickwork in the search for fresh wood and the ability to form such rhizomorphs
makes this a very destructive fungus. As long as there is a source of water
and nutrients, the rhizomorphs can transport these over considerable distances
and across inhospitable terrain to support the advancing mycelium. There is,
once again, a very apt military analogy. As long as there are both a well-stocked
supply depot and a secure, large-capacity supply line, a considerable front-line
force can be supported over a long distance.
), a species that can cause significant damage to timber in damp, poorly ventilated
buildings. The rhizomorphs produced by this fungus can extend for several metres
across brickwork in the search for fresh wood and the ability to form such rhizomorphs
makes this a very destructive fungus. As long as there is a source of water
and nutrients, the rhizomorphs can transport these over considerable distances
and across inhospitable terrain to support the advancing mycelium. There is,
once again, a very apt military analogy. As long as there are both a well-stocked
supply depot and a secure, large-capacity supply line, a considerable front-line
force can be supported over a long distance.
In all of the above examples, the rhizomorphs have been more-or-less at ground
level, but they can also be found off the ground. In tropical rainforests a
considerable amount of the leaf and twig litter that falls from the taller trees
is often trapped, at least temporarily, in understorey trees and shrubs. Numerous
wiry rhizomorphs (to 1mm in diameter) wind their way through the trapped litter.
A study in Ecuador found 200 to 300 kg (dry weight) of such trapped litter per
hectare, containing at least 500 to 600 metres of rhizomorph per kilogram of
litter. Another way of reporting that rhizomorph abundance is to say that there's
at least 100 kilometres of rhizomorph per hectare. A number of common rhizomorph-producing
fungi were in the genera Marasmius or Marasmiellus. Many of the
species in these genera produce tiny mushrooms as fruiting bodies, with the
caps often under a centimetre in diameter. The rhizomorphs hold on to fallen
leaves and twigs by producing splayed-out adhesion pads. Once these pads have
a secure hold on the plant debris, hyphae can penetrate and fan out as a more
diffuse mycelium to extract the nutrients inside. You can find out a bit more
about this study in the reference given in the button. ![]() Incidentally,
the author also reported that the rhizomorphs are so common that they are used
in Hummingbird nests - which can later produce crops of tiny mushrooms.
Incidentally,
the author also reported that the rhizomorphs are so common that they are used
in Hummingbird nests - which can later produce crops of tiny mushrooms.
Fairy rings
As already noted, a mycelium growing in the ground does not expand as a disk but as a ring, with most of the activity near the outer edge of the ring. Not surprisingly, mushrooms often appear to grow in a ring, the so-called fairy ring that reflects the underground mycelial presence. The most commonly seen rings are probably those produced by Marasmius oreades (which even gets the common name of Fairy Ring Mushroom). The mushrooms produced by this fungus do not have a striking appearance, generally being pale brown and often only 2 or 3 centimetres in diameter (though sometimes up to 5 centimetres). However, the mushrooms often appear in large numbers, in obvious rings, in open grassy areas such as sports fields and lawns in homes and public parks. These pictures show both the mushrooms in close view and views of a couple of fairy rings.
 Marasmius oreades |
 Marasmius oreades fairy rings |
Marasmius oreades is a widespread species, found in numerous countries in both hemispheres, and may appear at almost any time of the year (even summer) after some rain. While this may be the most commonly seen ring-forming species, several species of Agaricus (the genus that includes the Field Mushrooms) as well as a few puffball and puffball relatives are also fairly often seen in rings. While the rings produced by these fungi are often easily noticed because they often occur in open, grassy areas, many other fungi are known to produce their fruiting bodies in rings. However, it's easy to miss the ring arrangement in a bushy forest area.
The rate of growth of fairy rings varies, even within the one species. For example, several European studies of Marasmius oreades fairy ring growth showed a variety of average annual radial growth rates, from ten centimetres per year to thirty-five centimetres per year.
 In
some cases the ring-like underground mycelium has little or no visible effect
and only makes its presence known when it produces the visible fruiting bodies.
On the other hand, Marasmius oreades is an example of a fungus where
the mycelium has a visible effect throughout the year, often showing up as a
ring of brownish or dead grass, with two zones of greener, more vigorous grass
immediately inside and outside the ring of dead grass. The following diagram
shows a stylized aerial view of such a ring.
In
some cases the ring-like underground mycelium has little or no visible effect
and only makes its presence known when it produces the visible fruiting bodies.
On the other hand, Marasmius oreades is an example of a fungus where
the mycelium has a visible effect throughout the year, often showing up as a
ring of brownish or dead grass, with two zones of greener, more vigorous grass
immediately inside and outside the ring of dead grass. The following diagram
shows a stylized aerial view of such a ring.
What is the reason behind these zones of differing grass growth? The advancing edge of the underground mycelium secretes chemicals that break down organic matter in the soil to release nutrients for the fungus to use. For a short time the grass just beyond the outer edge of the mycelium can also make use of these nutrients, in particular nitrogen - which makes the grass much greener than the grass further ahead of the outer edge of the mycelium. A little way behind the outer edge, the mycelium is much denser, actively feeding on the liberated nutrients and leaving little for the grass. Moreover, the mycelium may be impervious to water, thereby reducing the water available to the grass and so putting further stress on the grass. Further inwards, there are the remnants of the cannibalized mycelium. This is broken down by various micro-organisms and the resulting nutrients are released into the soil. This increase in nutrients behind the mycelial ring explains the enhanced grass growth immediately behind the ring of brownish grass. Therefore the brownish grass recovers as the ring expands. The second diagram shows a vertical view, into the soil, along the dotted blue line in the first diagram.
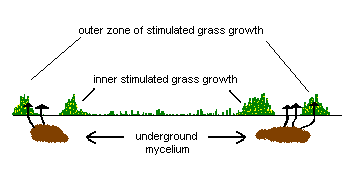
Many of the fungi that form fairy rings are saprotrophic species but mycorrhizal
fungi can also form fairy rings. In the case of a mycorrhizal fungus, the mycelium
is always attached to the roots of one (or more) plants. Such a fairy ring is
called a tethered ring whereas the rings formed by the unattached (non-mycorrhizal)
mycelia, such as those of Marasmius oreades, are called free rings.
The photo ![]() shows a tethered ring formed by a species of Cortinarius in a Eucalyptus
forest at Tidbinbilla Nature Reserve, in the Australian Capital Territory.
shows a tethered ring formed by a species of Cortinarius in a Eucalyptus
forest at Tidbinbilla Nature Reserve, in the Australian Capital Territory.
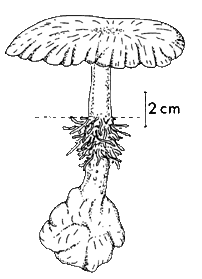 Lignosus rhinocerus from 'A Preliminary Polypore Flora of East Africa' (by permission of Leif Ryvarden) |
Sclerotia
A sclerotium is a mass of compacted hyphae, commonly more or less spherical
in shape and often with a dark, tough outer skin. Sclerotia are produced by
many fungi and range in size from under a millimetre to over 20 centimetres
in diameter. A sclerotium is a resting structure that allows a fungus to sit
out the hard times, analogous to (but clearly different from) a plant bulb or
corm. The outer skin protects the internal hyphal mass from drying out. When
conditions improve, the sclerotium may produce a new mycelium or fruiting bodies.
Two examples are Hypholoma tuberosum ![]() and Laccocephalum mylittae
and Laccocephalum mylittae ![]() ,
formerly called Polyporus mylittae. The sclerotium of Laccocephalum
mylittae has been used as a food by the Australian aborigines. Species in
the genus Morchella
,
formerly called Polyporus mylittae. The sclerotium of Laccocephalum
mylittae has been used as a food by the Australian aborigines. Species in
the genus Morchella ![]() are further examples of sclerotium-producing fungi.
are further examples of sclerotium-producing fungi.
This drawing shows a tropical polypore Lignosus rhinocerus. The dotted line represents ground level and you can see that the fruiting body has grown out from a buried sclerotium.
Sclerotia have been described as similar to the noduli, the primordial swellings that give rise to the fruiting bodies. Indeed one mycologist has written that sclerotia could be described as arrested and sometimes greatly oversized noduli. A pseudosclerotium is a heterogeneous mix of hyphae and substrate material. Sometimes the distinction can be difficult:
Pseudosclerotia begin their development by delimitating a usually rather large volume of substrate, wood or soil, by surrounding it with dense hyphal strands or sheaths. In most cases, hyphae grow out of the sheath into the delimitated chunk of substrate and slowly transform the latter into an intimate mixture of fungal biomass and residual substrate. When this happens in wood, such as felled trees or living roots, the wood inside the pseudosclerotium is protected against decay by other microorganisms and serves as a food reserve. In Wolfiporia extensa a sclerotium first grows around a root, then penetrates into it, and the resulting intermediate stage is a pseudosclerotium. Then the wood is completely digested and replaced by fungal mass. When this happens, the resulting body is a sclerotium. This means that the proportion of substrate in a pseudosclerotium varies enormously, from almost zero to more than half the body. Therefore the limit between sclerotia and pseudosclerotia is often blurred
.
The pseudosclerotium of Polyporus tuberaster, a northern hemisphere species, was long a puzzle, for here was something, apparently a stone, that produced fungi. It was noted in medieval times by Mathaeus Silvaticus, who referred to it as lapis lincis (literally 'lynx stone') in his pharmacopeia, Pandectarum medicinae. He wrote that it was created from the coagulation of lynx urine and noted that it could be used to produce fungi throughout the year. Be aware that various Classical authors refer to lynx stones but the evidence points to these being references to amber, not to sclerotia or pseudosclerotia. After Mathaeus' time the pseudosclerotium was known by various names, incorporating at least one of 'lynx' or 'stone' and three examples are Lapis lyncurius, Fungus lyncurius and Pietra fungaja. The first to publish a good study of this pseudosclerotium was Pier Antonio Micheli, in his 1729 monograph Nova plantarum genera, and this PDF ![]() presents both his illustrations and his words. The first modern examination of this fungus was published in 1845 by Carl Brunner and this PDF
presents both his illustrations and his words. The first modern examination of this fungus was published in 1845 by Carl Brunner and this PDF ![]() contains one of Brunner's plates showing several polypores growing from a pseudosclerotium.
contains one of Brunner's plates showing several polypores growing from a pseudosclerotium.
Dendropolyporus umbellatus, another northern hemisphere polypore, produces dense clusters of fruiting bodies from pseudosclerotia which can be coralloid or very contorted in shape, as shown in this PDF ![]() .
.
Collybia tuberosa is a sclerotium-producing mushroom genus, also found in the northern hemisphere. The mushrooms develop from the decayed remains of other fungi and a hand-coloured, 1820s illustration (as well as some additional information) is included in this PDF ![]() .
.
Longevity
In principle, a mycelium that can spread through the ground could keep on growing
indefinitely and large, old mycelia are known. Of course, once a mycelium has
spread to a considerable size it becomes more likely that it will suffer disruptions
and fragmentation. For example, a landslip, local flooding or antagonistic organisms
could destroy sections of the mycelium, leaving isolated fragments where once
there was a continuous mycelium.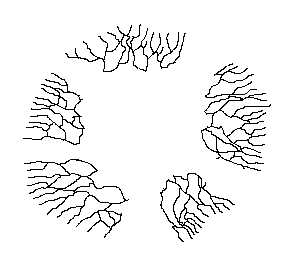
This is illustrated by the diagram, which is simply a copy of an earlier diagram, with several parts now erased to mimic disturbances of one kind or another. The five parts of the original mycelium can continue growing independently but, though no longer physically connected, they are still genetically identical and can be looked at as the one individual. As they continue to grow it is possible that at some future stage two of these currently disconnected mycelia will come into contact. Then, since genetically identical mycelia can fuse, the two mycelia can re-join.
Genetic testing answers the question of whether two separated mycelia are the
one individual and in this way various large, old mycelia have been found. An
example of large size and great age is provided by an Armillaria ostoyae
mycelium in Oregon. Armillaria produces mushrooms as fruiting
bodies and the genus is present in all continents, though with different species
in different areas. The species of Armillaria are plant parasites and
many species spread from tree to tree via rhizomorphs that grow through the
soil. The particular Armillaria ostoyae mycelium in question is spread
over 890 hectares and is estimated to be at least 2,400 years old. While this
individual may hold the size/age record, there are other documented instances
of fungal mycelia with impressive sizes and ages - when compared to the human
scale. ![]() Mycelia
spread over several tens of hectares, and with estimated ages of several hundred
to a thousand years, are not rare.
Mycelia
spread over several tens of hectares, and with estimated ages of several hundred
to a thousand years, are not rare.
On the other hand, consider species such as Cordyceps gunnii ![]() and Poronia erici
and Poronia erici ![]() .
The first is a parasite of larvae of Ghost Moths in the genus Oxycanus
and the second is a specialist dung fungus, found in the dung of various herbivores.
A couple of things in common to these two species is that their substrates are
very limited in size and separated in space. For example, while there are many
Oxycanus individuals in Australian woodlands or forests, they are not
in constant physical contact with one another. Similarly, there are numerous
piles of herbivore droppings in Australian grasslands, woodlands and forests
- but they are separated from one another. Once the Cordyceps mycelium
has spread throughout its host and extracted all the nutrients - it has nowhere
else to go. Similarly, the Poronia mycelium has nowhere to go once it
has spread through the dung. These species do not produce hyphae or rhizomorphs
that will spread through the soil looking for other larvae or dung piles. Once
the nutrients are exhausted these species produce fruiting bodies and rely on
spores. In the case of Cordyceps, any spores that get into fresh larvae
will start a new cycle of infection. Poronia spores that land on nearby
grass blades will be eaten by herbivores and later pass out in their droppings.
.
The first is a parasite of larvae of Ghost Moths in the genus Oxycanus
and the second is a specialist dung fungus, found in the dung of various herbivores.
A couple of things in common to these two species is that their substrates are
very limited in size and separated in space. For example, while there are many
Oxycanus individuals in Australian woodlands or forests, they are not
in constant physical contact with one another. Similarly, there are numerous
piles of herbivore droppings in Australian grasslands, woodlands and forests
- but they are separated from one another. Once the Cordyceps mycelium
has spread throughout its host and extracted all the nutrients - it has nowhere
else to go. Similarly, the Poronia mycelium has nowhere to go once it
has spread through the dung. These species do not produce hyphae or rhizomorphs
that will spread through the soil looking for other larvae or dung piles. Once
the nutrients are exhausted these species produce fruiting bodies and rely on
spores. In the case of Cordyceps, any spores that get into fresh larvae
will start a new cycle of infection. Poronia spores that land on nearby
grass blades will be eaten by herbivores and later pass out in their droppings.
In conclusion
The English mycologist Alan Rayner has spent many years studying the behaviour of mycelia in forests. In the introduction to the reference given in the button, he wrote:
"...there is perhaps a natural tendency to regard ...the mycelium as a boring, uniformly absorbent mass of hyphal threads which, whilst important in energy capture, only really become interesting when parts of it aggregate and differentiate into a fruit body...... I have increasingly come to regard the mycelium as a heterogenous army of hyphal troops, variously equipped for different roles and in varying degrees of communication with one another. Without a commander, other than the dictates of their environmental circumstances, these troops organise themselves into a beautifully open-ended or indeterminate dynamic structure that can continually respond to changing demands. Recall that during its potentially indefinite life, a mycelial army may migrate between energy depots; absorb easily assimilable resources such as sugars; digest refractory resources such as lignocellulose; mate, compete and do battle with neighbours; adjust to changing microclimatic conditions; and reproduce."
![An Australian Government Initiative [logo]](/images/austgovt_brown_90px.gif)

